All About The Atom
It is important to understand that:
all atoms are made of protons, neutrons, and electrons; and
all molecules and compounds are made of atoms.
Protons, neutrons, and electrons make atoms; atoms make molecules and compounds. Everything in the world is made of molecules and compounds. So, everything is made of atoms. And all atoms are just combinations of protons, neutrons, and electrons.
the basic unit of all matter.
the number of protons in an atom.
matter that has more than one element in it.
a very small part of an atom. Electrons have a negative charge. They exist in a cloud around the nucleus of atoms.
a basic kind of matter. Atoms with the same number of protons are all the same element.
an atom that has does not have the same number of protons and electrons. The atom either lost electrons or gained electrons. Ions have electromagnetic charge.
an atom with more or less neutrons than is normal for that element.
everything you can touch, feel, see. Real physical things in the world (and the universe) are all made of matter.
atoms joined together. Can be two or more of the same element, or different elements.
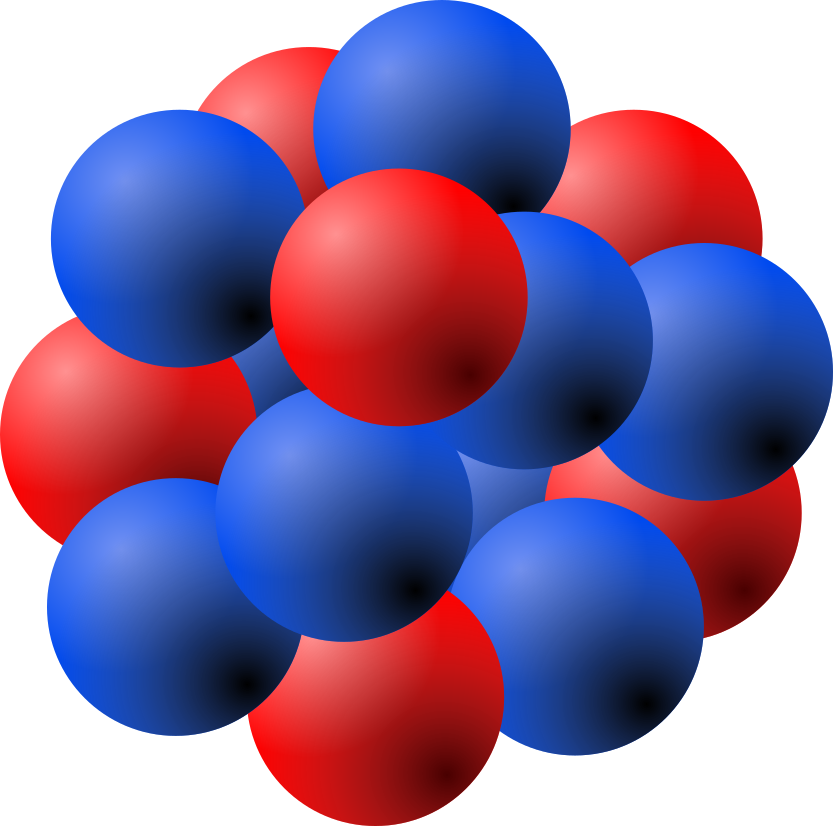
a small part of an atom. Neutrons are neutral — that is, they have no charge. They are in the nucleus of atoms.
the “ball” at the center of an atom. Protons and neutrons are in the nucleus.
the table that shows the elements in an organized way
a small part of an atom. Protons have a positive charge. They are in the nucleus of atoms.
Atoms are the basic unit that make up all matter. Everything you can see, touch, or smell is made up of atoms. There are many different types of atoms. These different atoms are called elements. For example, hydrogen, carbon, and gold are elements. The elements are organized on the periodic table.
Atoms are very, very, very small. Individual atoms are impossible to see without special tools.
Atoms join together to make molecules: for example, two hydrogen atoms and one oxygen atom combine to make a water molecule. When atoms join together it is called a chemical reaction.
Atoms are always moving. In solids, the atoms do not move very much. In solids, the atoms are tightly packed next to each other so they vibrate, but do not move around freely. Solids do not change shape easily because the atoms are held together strongly. Clothes, paper, pencils, and cars are all examples of solids.
In liquids, the atoms are loosely held together, but are free to move around a little. Liquids easily change shape but still stay together more than gases do. Water and milk are examples of liquids.
In gases, the atoms are free to move around. They do not stay together and they don’t really have a shape. The air in a room is an example of a gas. Another example is steam from boiling water.
Atoms change from solid form to liquid to gases based on how much energy they have.1 This is why you can make steam by putting ice in a cooking pot and turning on the stove. The heat from the stove will give the ice enough energy to change it from a solid (ice) to a liquid (water). Eventually the energy from the stove will turn the water into a gas (steam).
Every atom is made up of three kinds of smaller particles, called protons, neutrons, and electrons. Protons are positively charged. Neutrons are “neutral”, that is, they have no charge. Electrons are negatively charged. The protons and neutrons are heavy and stay together in the middle of the atom. This is called the nucleus. The nucleus is surrounded by a cloud of electrons. Electrons have very little mass. Because they have a negative charge, they are attracted to the positive charge of the nucleus. This attraction is called electromagnetic force.

Most atoms have a neutral charge. When the number of protons (positive) and electrons (negative) is the same, the charges cancel out. However, in ions (atoms that have lost or gained electrons) this is not always the case. Ions will have either a positive or a negative charge.
Ions are one way that atoms form compounds. A negative ion will be attracted to a positive ion. Together, the two (or more) atoms can have zero charge.
The number of protons an atom has tells us what element it is. Hydrogen, for example, has one proton. The element sulfur has 16 protons. Any atom with only one proton will be hydrogen. Any atom with exactly 16 protons will be sulfur.
Atoms of the same element can have different numbers of neutrons. These are known as isotopes of the element. All isotopes of an element are still that element. A hydrogen atom (one proton) with one neutron is still hydrogen. A hydrogen atom with zero neutrons is still hydrogen. Changing the number of neutrons does not change what element an atom is. Changing the number of protons does change what element the atom is. In normal everyday life people cannot change the number of protons an atom has.
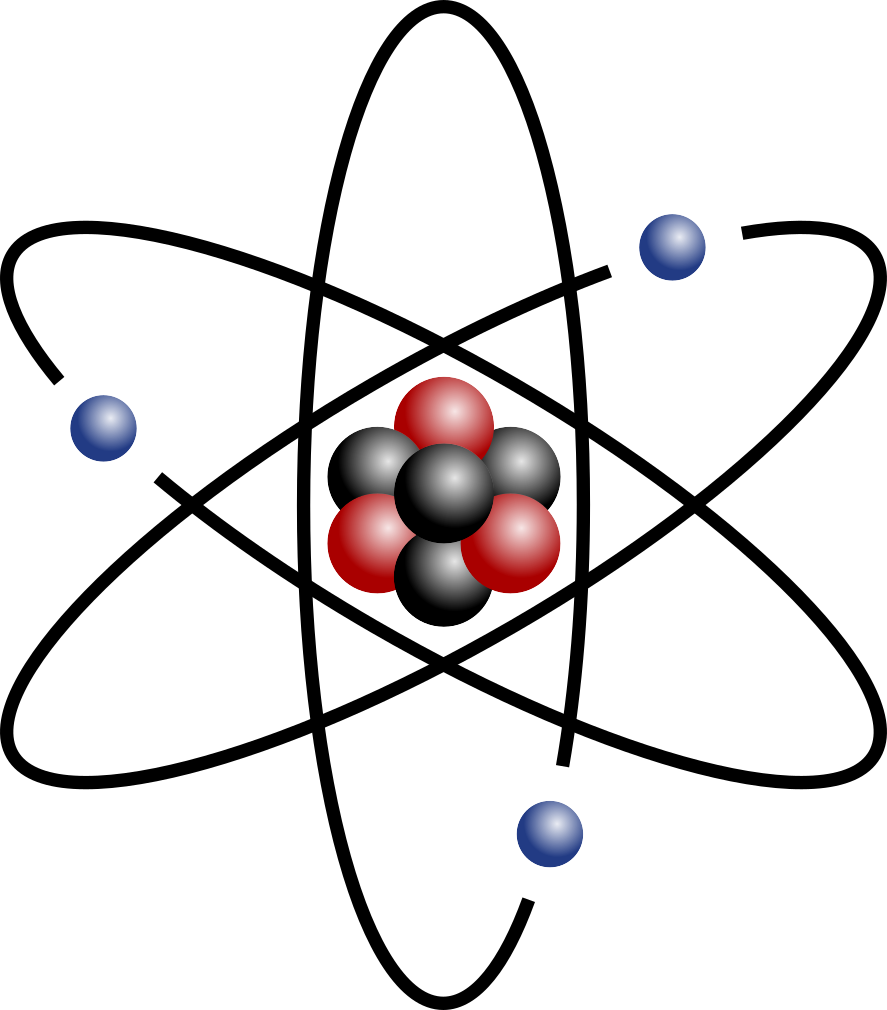
Electrons orbit, or travel around, the nucleus. We call this the atom’s electron cloud. Electrons are attracted to the nucleus because of the electromagnetic force. Electrons have a negative charge and the nucleus has a positive charge, so they attract each other.
In the electron cloud, some electrons are further out than others. They are in different levels. These are called electron shells or valence levels. In most atoms the first level can hold two electrons, and all the shells after that can hold more, but are often “full” if they have eight electrons. Electrons will fill a level before starting a new level further out. The last level will usually not be full (will not have 8 electrons in it). When an atom has a full outer valence level, it is stable and will not react with other atoms easily.
Electrons can only be in the set valence levels. They cannot be just anywhere in the space around the nucleus.
Valence levels are another way that atoms can form compounds. Atoms can “share” the electrons in their outermost valence levels (the levels farthest out that have any electrons). When they share electrons like this, both atoms can have full outer levels. This makes them more stable, which is why the atoms stay together as a compound.
The information below is not necessary to understand the basics of atoms. But, it can help you understand how science progresses.
The word “atom” comes from the Greek “atomos”. The Greek philosopher Democritus was the first person (we know of) to say that everything is made of atoms. He lived around 400 B.C.E. He basically had a lucky guess. He had no way of proving his hypothesis.
In 1777, a French chemist defined the term element for the first time. He said that an element was any basic substance that could not be broken down into other substances by chemists. Any substance that could be broken down was a compound.
In 1803, an English philosopher said that elements were tiny, solid balls made of atoms. He thought that compounds are combinations of elements. He also said that in compounds, the atoms always combine the same way.
In 1869, the scientist Dmitri Mendeleev published the first version of the periodic table. The periodic table groups elements by their atomic number. Elements in the same column usually have similar properties. For example, the elements in the column with helium and neon have similar properties. All these elements are gases. None of them have any color or smell. Also, they are unable to combine with other atoms to form compounds.
The physicist J.J. Thomson discovered the electron in 1897. He realized they had a negative charge, unlike protons (positive) and neutrons (no charge). Thomson created the plum pudding model,2 which said that an atom was like plum pudding: the dried fruit (electrons) were stuck in a mass of pudding (protons).
In 1909, however, Ernest Rutherford proved that most of the mass of an atom is in a very small space called the atomic nucleus, or just the nucleus. Every atom is mostly empty space!
In a famous experiment, Ernest Rutherford shot alpha particles3 at gold foil. Gold is much heavier than alpha particles, but the alpha particles were moving very fast. If Thomson’s plum pudding model was correct, the alpha particles should slow down, stop, or maybe bounce off of the gold.
Instead of stopping, many of the alpha particles went straight through the gold foil without slowing down. This proved that atoms are mostly empty space. Some particles were deflected a little bit before they came out—they did not go straight through, but hit something before coming out behind the foil.
Amazingly, some alpha particles bounced straight back at Rutherford! The only explanation that made sense was: there was a small area of mass and lots of empty space in the gold atoms. The alpha particles must have hit an atom’s nucleus straight on and bounced right back the way they came! This proved that the mass of an atom was mostly in its nucleus. Therefore, the plum pudding model could not be correct.
In 1913, Niels Bohr introduced the Bohr model. This model showed that electrons travel around the nucleus in circular orbits. This was more accurate than the Rutherford model. However, it was still not completely right. The most accurate model so far comes from Edwin Schrödinger. He realized that the electrons exist in a cloud around the nucleus.
Later in the 20th century, physicists showed that protons and neutrons are actually made of other particles, called quarks. Luckily, we don’t have to get into that right now. We can pretend that protons, neutrons, and electrons are the basic building blocks of matter. If you take chemistry or physics later in high school or college, you will learn more about quarks.
This short history of the atom shows how science works. The first thoughts were just guesses. As technology and people’s understanding of chemistry got better, people made hypotheses and then tested them. The data from the new experiments gave scientists ideas for new hypotheses and new experiments to test those hypotheses. Sometimes scientists had to wait for new technology before they could do better experiments. Each round of hypotheses and experiment gave scientist a better idea of what atoms are. Or, sometimes, experiments would disprove an idea and at least show what atoms are not. In both cases, people’s understanding of atoms improved.
As we mentioned above, we already know that the protons, neutrons, and electrons model is not the most accurate. One day new experiments and new technology may change our understanding of quarks also. However, for a lot of what we do in the real world with atoms, the proton, neutron, and electron model works to explain things 100% of the time. Elements and compounds act like they are made of protons, neutrons, and electrons interacting. At that level, new information refines the details of the model, but does not replace it.
This reading was compiled and modified from the following sources:
at Simple Wikipedia. Accessed 2022 August 1. Published under the Creative Commons Attribution-ShareAlike 3.0 Unported License. https://simple.wikipedia.org/wiki/Atom
from Wikimedia Commons. Accessed 2022 August 1. Published under the Creative Commons Attribution-ShareAlike 3.0 Unported License. https://commons.wikimedia.org/wiki/File:Nucleus_drawing.svg
with three Bohr model orbits and stylised nucleus.svg from Wikimedia Commons. Accessed 2022 August 1. Published under the Creative Commons Attribution-ShareAlike 3.0 Unported License. https://commons.wikimedia.org/wiki/File:Stylised_atom_with_three_Bohr_model_orbits_and_stylised_nucleus.svg
This document is licensed under the Creative Commons Attribution-Share Alike 4.0 International license.
The original LaTeX files and images used to create this reading are
available at:
https://www.chrisspackman.com/educator-resources/readings/all-about-the-atom/all-about-the-atom-en-latex-files.zip